Adjunctive and Prophylactic Use of Antibacterial Agents in Dentistry
Course Number: 475
Course Contents
Inhibitors of Bacterial Cell Wall Synthesis
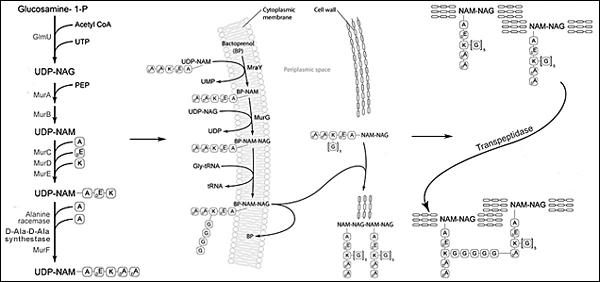
Most pathogenic bacteria have a cell wall that provides tensile strength and maintains intracellular osmotic pressure. Its synthesis progresses in three steps: (1) monomers are synthesized in the cytoplasm from amino acid and sugar building blocks; (2) Bactoperol transfers the monomers across the cytoplasmic membrane where they are polymerized into linear peptidoglycan chains; finally, (3) transpeptidase cross-links peptidoglycan chains into a three-dimensional mat (Figure 6).8
Figure 6. Bacterial Cell Wall Synthesis.
Modified from Kasmar AG, Hooper D. Pharmacology of bacterial infections: cell wall synthesis. In Golan DE, Tashjian, Jr. AH, Armstrong EJ, Armstrong AW. Ed. Principles of pharmacology. The pathophysiologic basis of drug therapy. 2nd ed. 2008. Wolters Kluwer/Lippincott Williams & Wilkins. Baltimore, MD.8
Step 1. Monomer synthesis | Step 2. Monomer polymerization | Step 3. Polymer cross-linking
A number of drugs inhibit cell wall synthesis. Most important are vancomycin, which targets monomer polymerization; and the β-lactams, e.g., penicillins and cephalosporins, which block polymer cross-linking.8,33-41 β-lactam antibacterial agents also activate autolysins. Autolysins punch holes in bacterial cell wall and disrupt its integrity.8 Transpeptidase antagonism and autolysis prevent bacterial self-maintenance, i.e., remodeling and repair; and replication.
Vancomycin
Vancomycin is bactericidal in susceptible organisms. It is primarily effective against aerobic gram-positive cocci and bacilli.8,38-40 It does have activity against some anaerobic gram-positive, but not against gram-negative bacilli. Since facultative and anaerobic gram-positive and gram-negative cocci and bacilli predominate in all types of odontogenic infections, Vancomycin does not have the requisite spectrum to be considered an *empirical* option in treating odontogenic infections.
Penicillins
Penicillins are bactericidal in susceptible organisms.8,34,37 Narrow-spectrum penicillin V potassium and broad-spectrum amoxicillin and amoxicillin with clavulanic acid have the requisite spectra to be considered as *empirical* options in treating odontogenic infections.11,42 However, neither narrow-spectrum nor broad-spectrum penicillins are active against β-lactamase producing bacteria; and certain β-lactamases produced by bacteria now confer resistance to clavulanic acid as well.8,43-51
Penicillin V potassium and amoxicillin formulations are not inactivated by gastric acid and also have the advantage that they may be given with meals. They are widely distributed to most tissues and body fluids, cross the placenta and they are excreted into breast milk. The penicillins undergo hepatic biotransformation. The metabolites and the unchanged fraction of the drugs are excreted rapidly in individuals with normal renal function.
Cephalosporins
The cephalosporins are bactericidal in susceptible organisms.8,33,35,36,52Most are primarily active against aerobic gram-positive cocci and bacilli. Second generation cephalosporins (e.g., cefaclor) have an overlapping spectra with those of penicillin V potassium and amoxicillin formulations and are more β-lactamase resistant than the first generation cephalosporins. However, cephalosporins, in general, offer no therapeutic advantage over penicillins as empirical options in treating odontogenic infections.